The Beginning. The Launch. Misusing the Media. The Scam is Busted. New Horizons | Valkee dislikes this | Valkee attacks / comment
NOTE: This page is outdated, it only covers things up to 2012. Important additions done only on the GLOOM blog.
In 2006 Nokia engineer Antti Aunio and biologist Juuso Nissilä applied for a Finnish patent for a device consisting of ear-plugs that could deliver LED light into the ear canals, and a small controller box. 1 According to Nissilä, the product idea was possibly born as early as 2005. 2

In spring 2007, their company Valkee Ltd. was founded to further develop (industry speak for to market) the device. 3 Valkee aimed to produce supporting evidence, a challenge for an invention unknown to the scientific community, made by non-medical outsiders, with a complete lack of basic research, not a single article ever written about the topic. They had to make the thing look like something scientific.
They convinced Anssi Vanjoki, Nokia's no.2, and later some venture capitalists, to invest into the start-up. 4 Valkee brought in some academics from the University of Oulu, who would later pose as experts. They had been on the payroll of pharmaceutical companies and had never published about the topic before.
 |
|---|
| Statement from the Medical Hypotheses article: "Hired guns" - professors for rent |
It was easy, however, to obtain authority approval for their medical device, which is available in Europe for useless or even potentially dangerous things. 5
The launch of the light headset in 2010 was accompanied by an intense PR campaign, the most notable event being the INNOSUOMI award presented by the Finnish President Tarja Halonen, acquired by Valkee with false claims. 6 The story of the innovative young firm from the north, promising help for the many sufferers of Finlands national disease, Seasonal Affective Disorder, hit the media like a bombshell. A profitable business with world-wide markets was exactly what the nation needed and wanted, at the time of Nokia's spectacular downturn which left thousands unemployed and the Finnish state whithout it's most important taxpayer.
At this point, Valkee could only cite a small unpublished open study. According to regulatory guidelines, other than controlled studies are not able to demonstrate any benefit other than the placebo effect. 7
Christmas however, did not bring great sales for Valkee, and their financial result was devastating. 8 In spring 2011, they were also scientifically bancrupt, as in their only completed placebo-controlled study which could have shown a real benefit sham treatment got the best results when compared to Valkee's ear light. 9 10
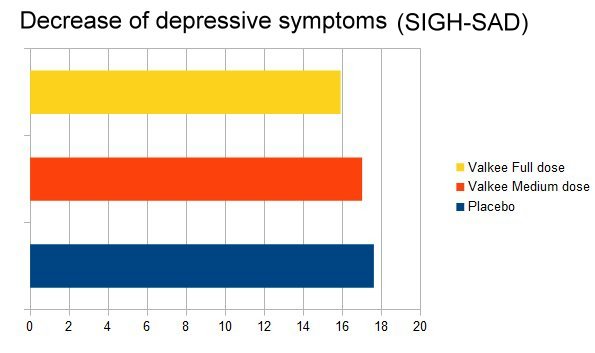 |
|---|
| Better off putting placebo in the ear: Results of Valkee's randomized controlled study |
The venture capitalist Timo Ahopelto rescued the struggling company with his Lifeline Ventures fonds and replaced Nissilä as CEO. 3 Valkee restarted it's media campaign with an intensity unseen before. Up to May 2011, Nissilä already counted more than 1000 appearances in TV, newspapers, journals, for the ear light treatment. 11 Ahopelto reported even an (one-word) appearance in a Finnish Donald Duck comic. The bigger impact, however, had the popular Fingerpori strip: It suggested to light up another orifice, for the same 199,95 price.
Nobody stood up to destroy the illusion, when Valkee announced dubious scientific revolutions in the press. The scam went unnoticed by the scientific community. Valkee provided just stuff that looked like science, enough to sell the fake story.
The majority of the public although, remained sceptic, but critics were silenced when Valkee announced a positive randomized controlled study in November 2011, making it into the TV evening news. 12 The results were clumsily falsified and the real numbers indeed showed the device to be an expensive placebo. 9 10 Nevertheless, no-one seemed to care and this opened the way for better sales on Christmas 2011.
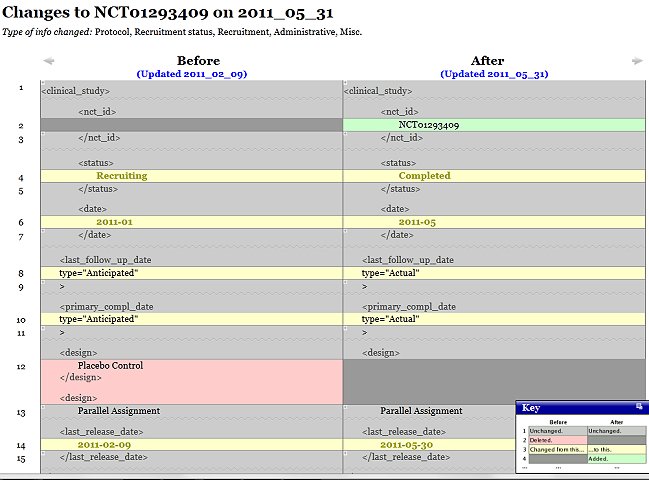 |
|---|
| Fixing unwanted results: Placebo vanishes as the trial completes (clinicaltrials.gov) |
Valkee, however, overdid the bad thing, and in February 2012 a whistleblower teamed up with two well-known finnish journalists, Renny Jokelin and Magnus Berglund (3-times Prix Europa nominee and famous for busting the Patria scandal). They made a piece for MOT, a TV program grossly comparable to the BBC's Panorama. It aired at primetime 8.00 pm on the first YLE channel, reaching a market share of 34%. 13
It effectively ended Valkee's media misuse overnight. The next day, Nissilä confirmed the key facts from MOT in a radio interview, but still claimed to have made a scientific revolution. 14 Valkee's freshly appointed CEO Pekka Somerto did not want to comment, only remarking that it was just what was to be expected from MOT.
Later in 2012, Valkee's scam won it's second big price: The HuuHaa (flim-flam) award by the finnish Skeptics for the marketing of the device without sufficient evidence. Two years earlier, the winner was the PowerBalance magnetic wristband. 15
Fortunate for them, the Finnish language is not widely understood internationally. Googling Valkee's claims only brings up their PR releases, and not that the scam was busted long ago in Finland.
Based on the traditional believe into authorities in Finland it was marketed by real professors, and TV! - and the fact that neither users nor journalists were able or willing to check the scientific base, Valkee tricked at least some 10.000 people into buying this expensive toy. Their markets are now the UK, Germany, even Japan.
There is still not a single peer-reviewed article supporting this "innovation", and strong evidence against it. Valkee is still selling an expensive, useless sham device for a real disease with possibly deadly complications that otherwise has to some extent effective treatment options. The scam continues.
Sources / References:
1 Finnish patent 121988B, issued on Jul 15, 2011, recieved first on Sep 6, 2006; PDF on file. -see picture-
2 Sari Okko w/ Juuso Nissilä: Valkee Oy - Light treatment for seasonal affective disorder. (undated) Foundation for Finnish Inventions website, retrieved 6.2.2013. Note that the product did not emerge from a scientific discovery and/or process, but from an idea "over a cup of tea".
3 Finnish trade register data, available at www.prh.fi, retrieved 6.2.2013.
4 (no authors named) Vanjoki sijoitti kaamosmasennusta ehkäisevään yhtiöön. Helsingin Sanomat website, 15.12.2010, retrieved 6.2.2013.
5 Deborah Cohen: How a fake hip showed up failings in European device regulation. BMJ 2012; 345:e7090.
6 Ministry of Employment and Economy: Uutiskirje (Newsletter) 15.12.2010, retrieved 6.2.2013.
7 The European Agency for the Evaluation of Medicinal Products: Note for Guidance on Clinical investigation of medicinal products in the treatment of depression. 25.4.2002, retrieved 6.2.2013.
8 Valkee Oy: Tasekirja (Balance sheet), 28.2.2011; PDF on file.
9 clinicaltrials.gov, register entry for NCT01293409. Identity of the study w/ ref. 10 was also confirmed by Valkee's Timo Ahopelto and is not disputed by Valkee Ltd. The placebo control was removed from the design after the trial's completion. -see picture-
10 Results in Jurvelin H, Jokelainen J, Nissilä J, et al: Transcranial Bright Light Treatment via Ear Canals in Seasonal Affective Disorder (SAD) - a Randomized Controlled Study (abstract). International Journal of Psychiatry in Clinical Practice, 2011; 15(Suppl 2): 27-8. PDF on file. Also available from Valkee's website. Note that the placebo results are presented as such for an active treatment.
11 Juuso Nissilä: public speech "Ideasta kasvuun" (from idea to growth) at the Enterprise Forum 2.0, 2/2011, University of Oulu, 17 May 2011. Video on file (was removed from the university's website after the MOT TV program aired).
12 YLE News (Uutiset), 20:30 on 8 Nov 2011, channel YLE 1.
13 MOT, 20:00 on 12 Mar 2012, channel YLE 1.
14 Juuso Nissilä: Interview w/ Radio Mega, 13.3.2012. MP3 on file.
15 YLE News: LED earbuds awarded humbug prize. 6.12.2012. Retrieved on 11.11.2013.

